A stable molecule has less energy than the same set of atoms separated one must provide at least this energy in order to take the molecule apart.

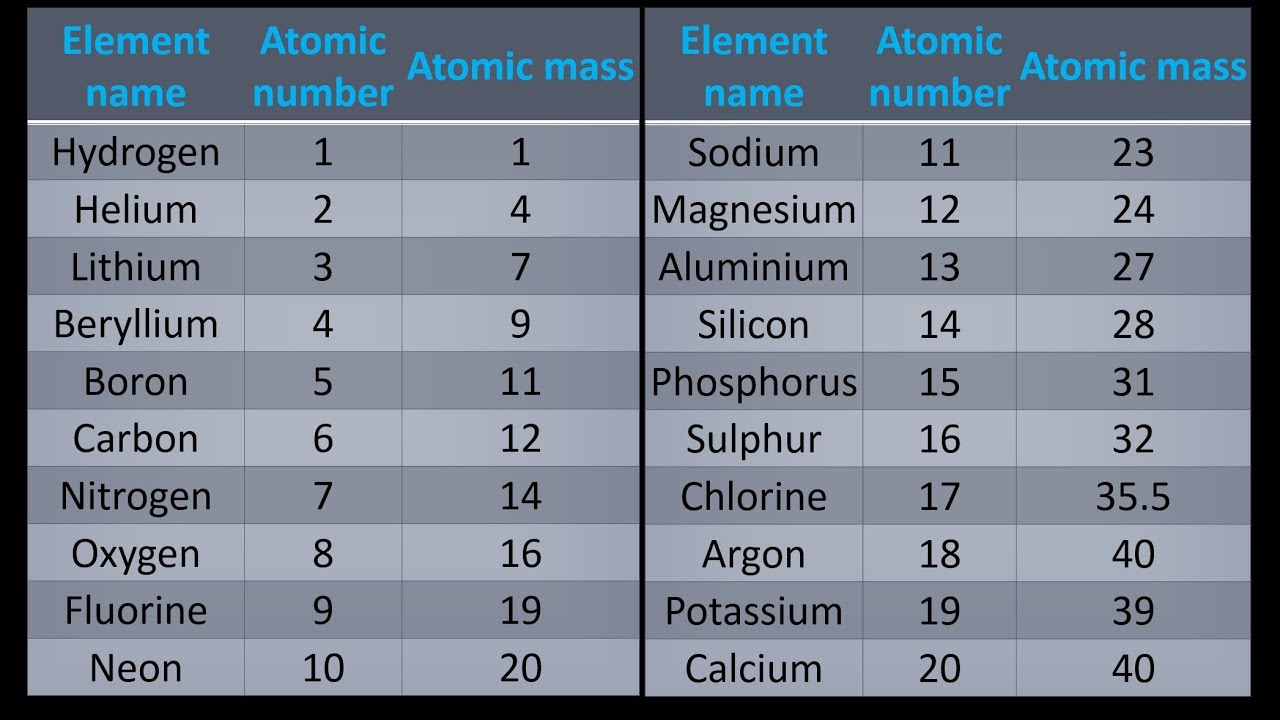
The structure and interactions of matter at the bulk scale are determined by electrical forces within and between atoms.The repeating patterns of this table reflect patterns of outer electron states. The periodic table orders elements horizontally by the number of protons in the atom’s nucleus and places those with similar chemical properties in columns.Each atom has a charged substructure consisting of a nucleus, which is made of protons and neutrons, surrounded by electrons.PS1.A: Structure and Properties of Matter Refine a solution to a complex real-world problem, based on scientific knowledge, student-generated sources of evidence, prioritized criteria, and tradeoff considerations.Construct and revise an explanation based on valid and reliable evidence obtained from a variety of sources (including students’ own investigations, models, theories, simulations, peer review) and the assumption that theories and laws that describe the natural world operate today as they did in the past and will continue to do so in the future.Apply scientific principles and evidence to provide an explanation of phenomena and solve design problems, taking into account possible unanticipated effects.(HS-PS1-7)Ĭonstructing Explanations and Designing SolutionsĬonstructing explanations and designing solutions in 9–12 builds on K–8 experiences and progresses to explanations and designs that are supported by multiple and independent student-generated sources of evidence consistent with scientific ideas, principles, and theories. Use mathematical representations of phenomena to support claims.Simple computational simulations are created and used based on mathematical models of basic assumptions. Mathematical and computational thinking at the 9–12 level builds on K–8 and progresses to using algebraic thinking and analysis, a range of linear and nonlinear functions including trigonometric functions, exponentials and logarithms, and computational tools for statistical analysis to analyze, represent, and model data. Using Mathematics and Computational Thinking Plan and conduct an investigation individually and collaboratively to produce data to serve as the basis for evidence, and in the design: decide on types, how much, and accuracy of data needed to produce reliable measurements and consider limitations on the precision of the data (e.g., number of trials, cost, risk, time), and refine the design accordingly.
:max_bytes(150000):strip_icc()/PeriodicTable-58b5d9253df78cdcd8d03ebb.jpg)
Planning and carrying out investigations in 9-12 builds on K-8 experiences and progresses to include investigations that provide evidence for and test conceptual, mathematical, physical, and empirical models.
Students who demonstrate understanding can:
